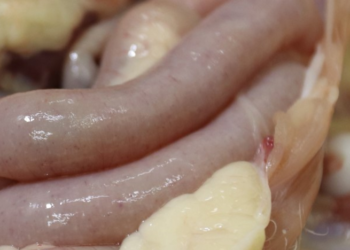
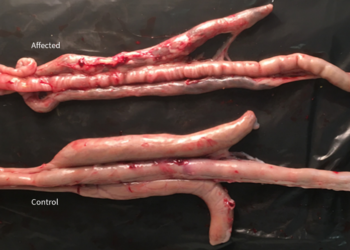
The global cost of coccidiosis has been recently estimated to be >$3 billion per annum. This cost calculation is based on direct production losses and indirect costs for prevention measures. However, most of this financial loss comes from an increased feed conversion ratio (FCR) and decreased weight gain (Williams, 1999). In addition, clinical disease – in broilers mainly caused by Eimeria tenella – leads to increased mortality. Finally, Eimeria is also one of the most important predisposing factors for bacterial enteritis.
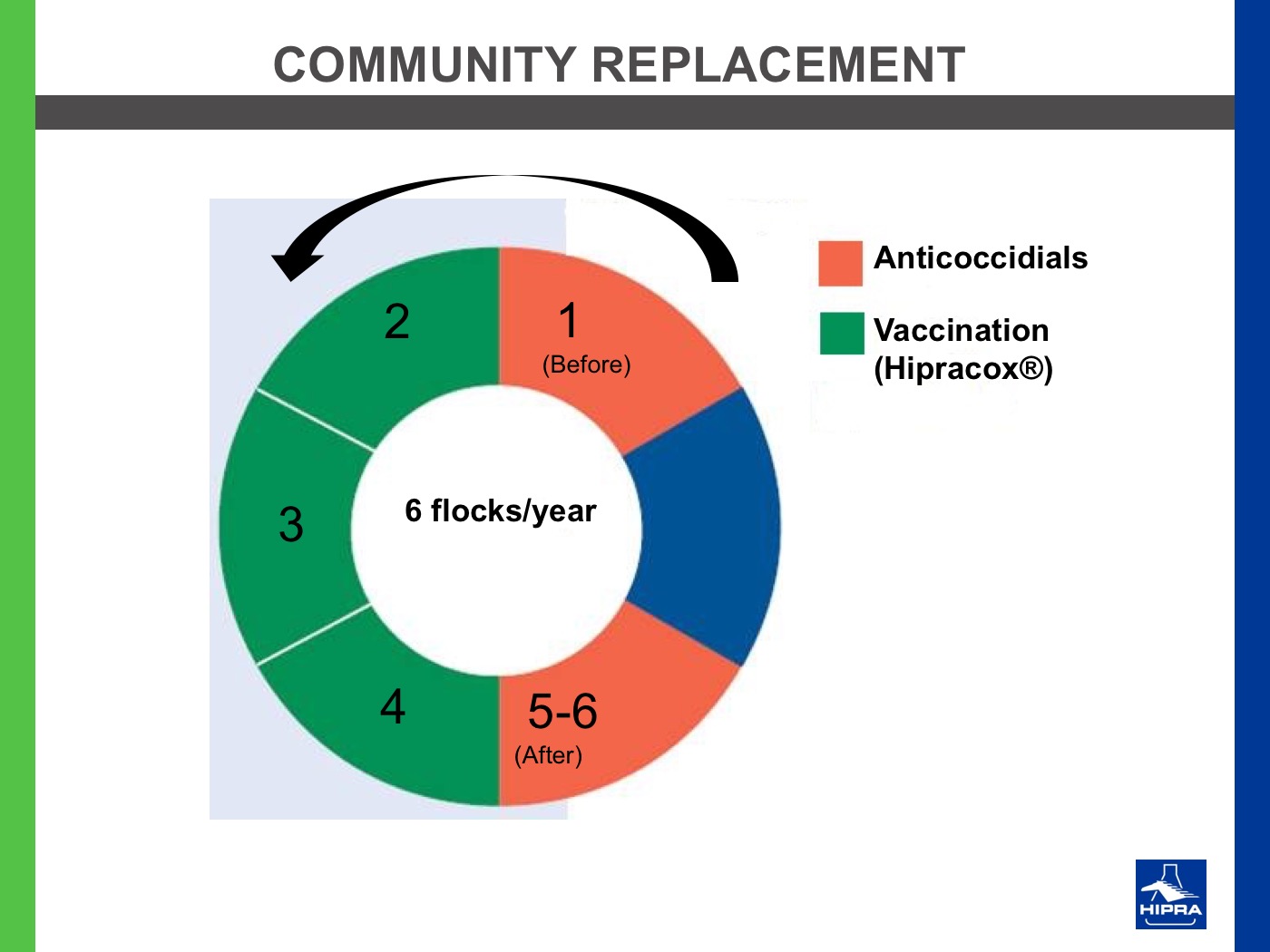

Anticoccidial feed additives or anticoccidial drugs have been and still are the most widely used prevention tool in broilers. These products have many merits but the two main issues associated with their use are (i) reduced sensitivity of Eimeria parasites when a certain product is used for too long or too often and (ii) cross resistance between certain compounds (Marien et al., 2007).
Recent regulatory developments – i.e. the reduction in withdrawal times of some monovalent ionophores and nicarbazin – have increased flexibility for broiler producers but also worsened the above-mentioned issues.
In fact, exposure of Eimeria to these compounds has increased – since they are used during the lifespan of a broiler flock – as has the use of products of the same category (monovalent ionophores), between which cross-resistance exists.
Therefore, there is an intensified need for alternative approaches to Eimeria prevention. Rotational programmes, where anticoccidial feed additives with different modes of action are used judiciously, are one such programme. Another increasingly popular approach is the regular use of Eimeria vaccines as part of a long-term prevention strategy.
In fact, the use of Eimeria vaccines as part of a rotation programme (alternating cycles with vaccination with cycles with anticoccidial programmes) has proved to restore the sensitivity of the Eimeria field population to anticoccidial drugs, as a consequence of the introduction of sensitive vaccine strains in the poultry houses (Chapman and Jeffers, 2015; Peek and Landman, 2011; Dardi et al., 2015).
In the study conducted by Dardi et al. (2015) the results indicate that three consecutive vaccinations changed the resistance profile of Eimeria spp. that populated the study farm and controlled the coccidiosis problems at least for two flocks after vaccination.
On the other hand, the 2nd cycle post-vaccination already showed the development of some resistance, indicating that three consecutive vaccinations may be insufficient to change the profile to full sensitivity. This could be because the farm had never been vaccinated before and for this reason Eimeria field strains had reached a marked level of resistance.

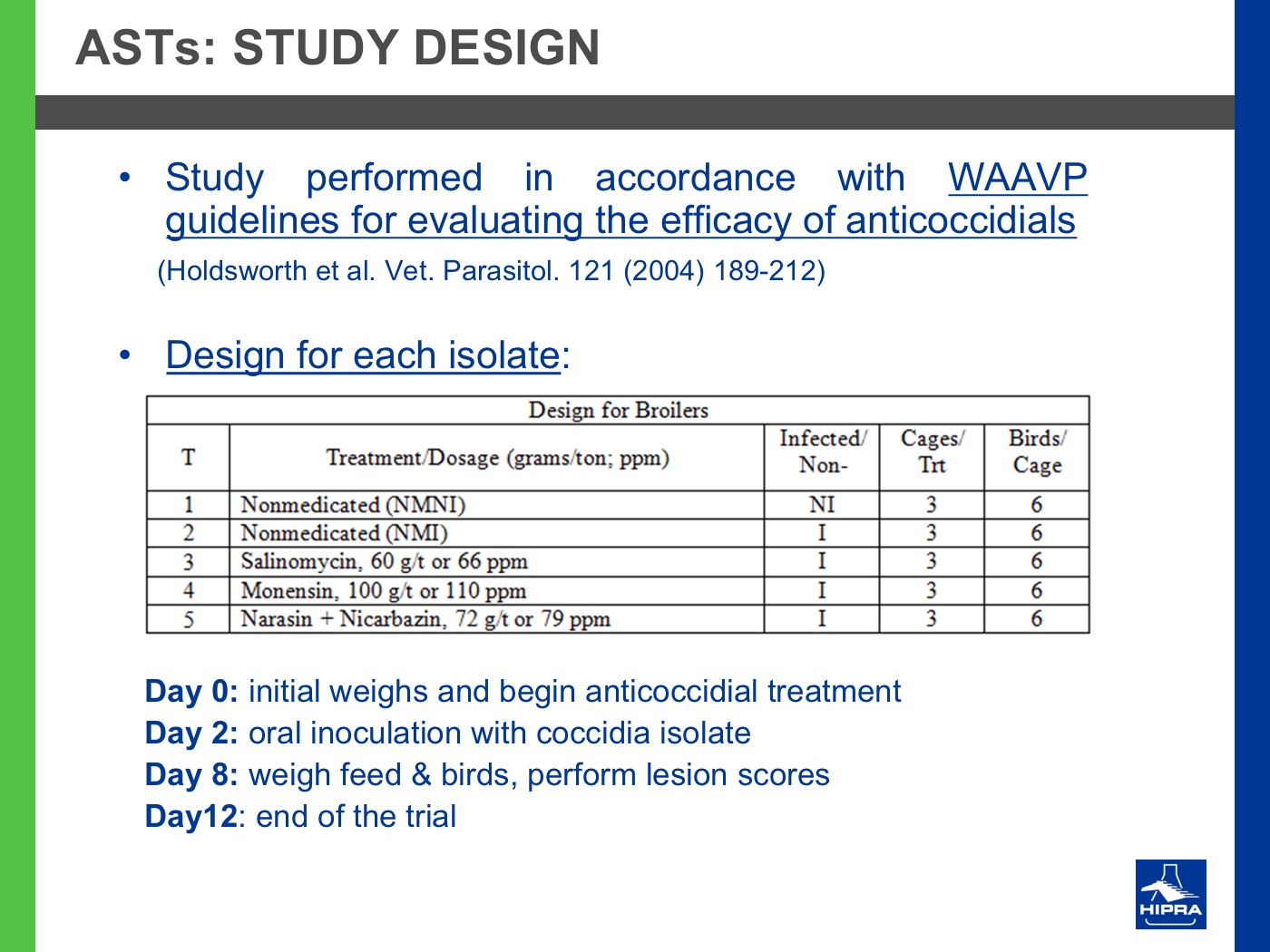
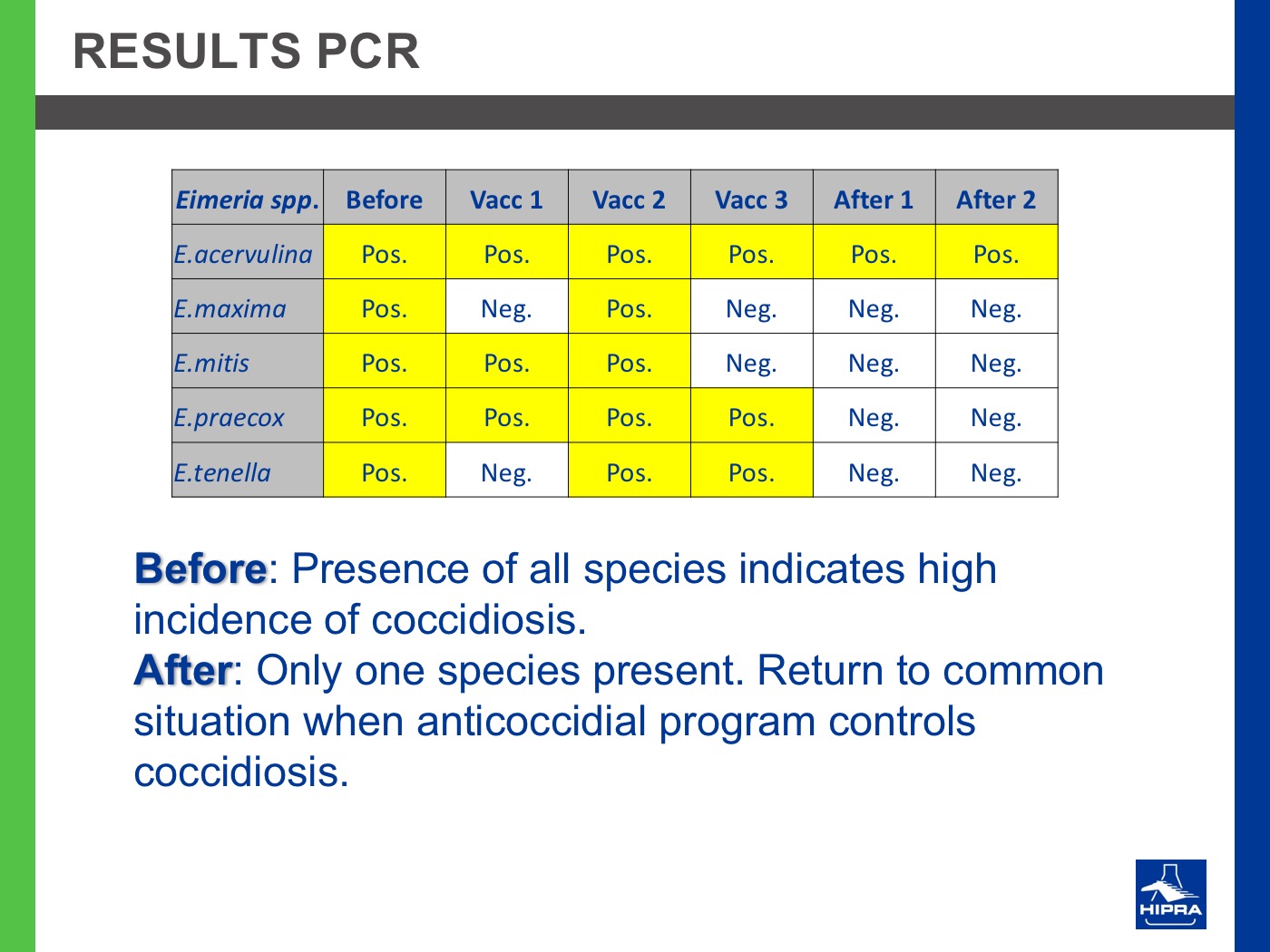
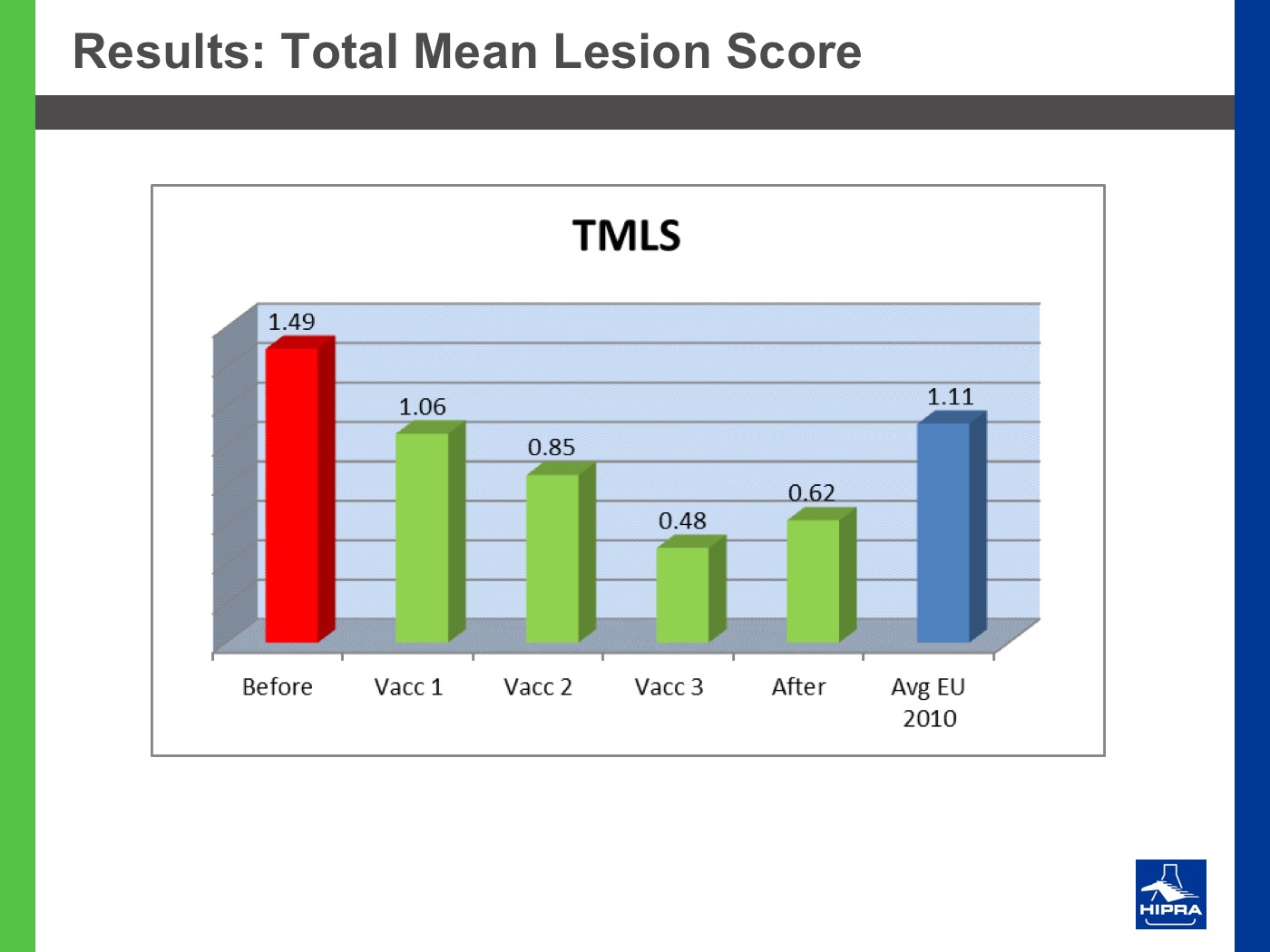
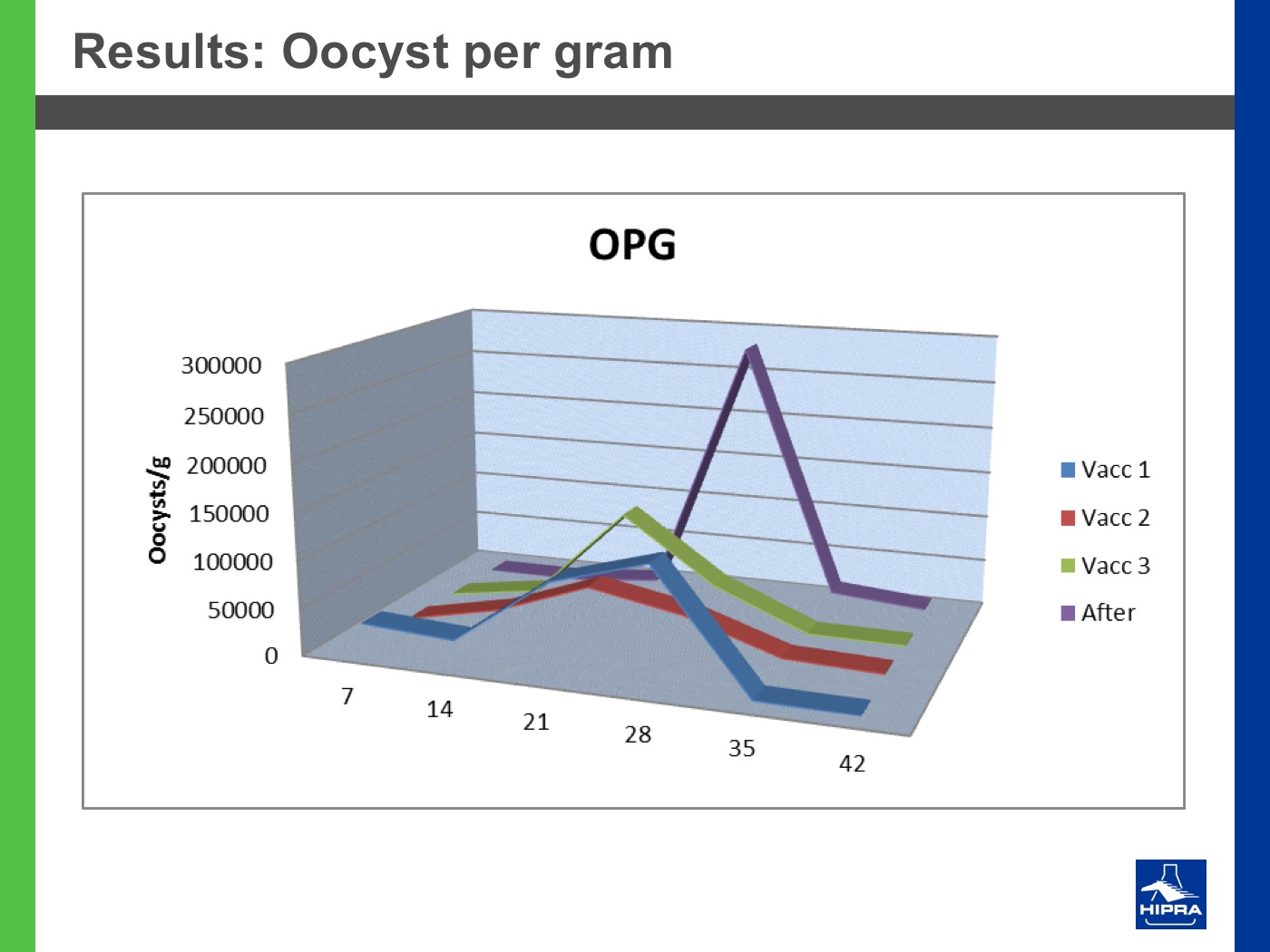

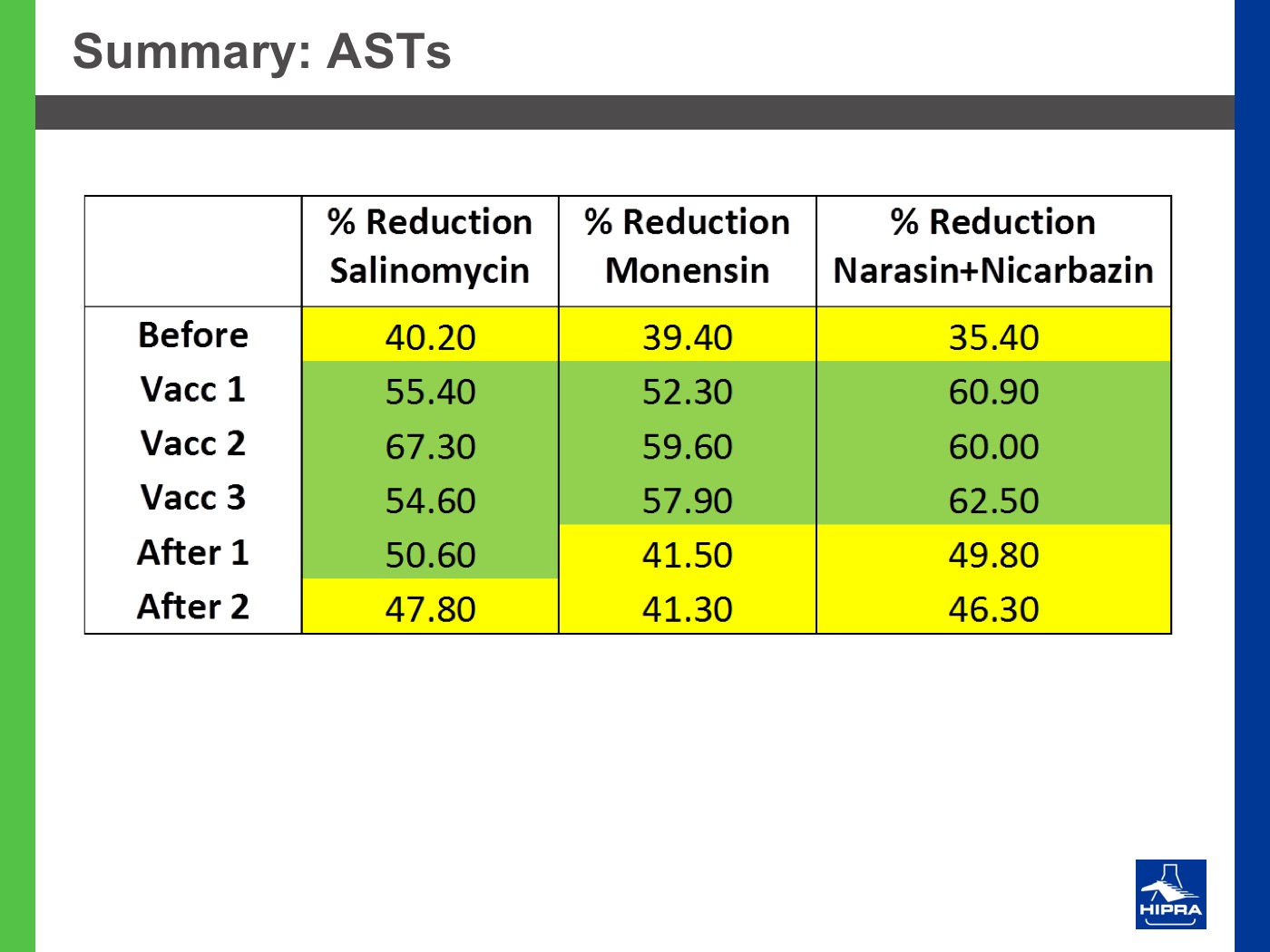
Additionally, the introduction of precocious attenuated Eimeria vaccines such as HIPRACOX® into rotation programmes (Ronsmans et al., 2015) showed that comparing the productive parameters before, during and after vaccination the following tendencies were detected:
- In general, there were never statistically significant differences between productive results before and during vaccination, shattering the popular belief that it is hard to obtain similar zootechnical results when preventing coccidiosis by vaccination, compared to using anticoccidial feed additives.
- Flocks after vaccination always showed statistically significantly improved FCRs and EPEFs when compared to the cycles before vaccination, whereas numerically results always improved after vaccination.

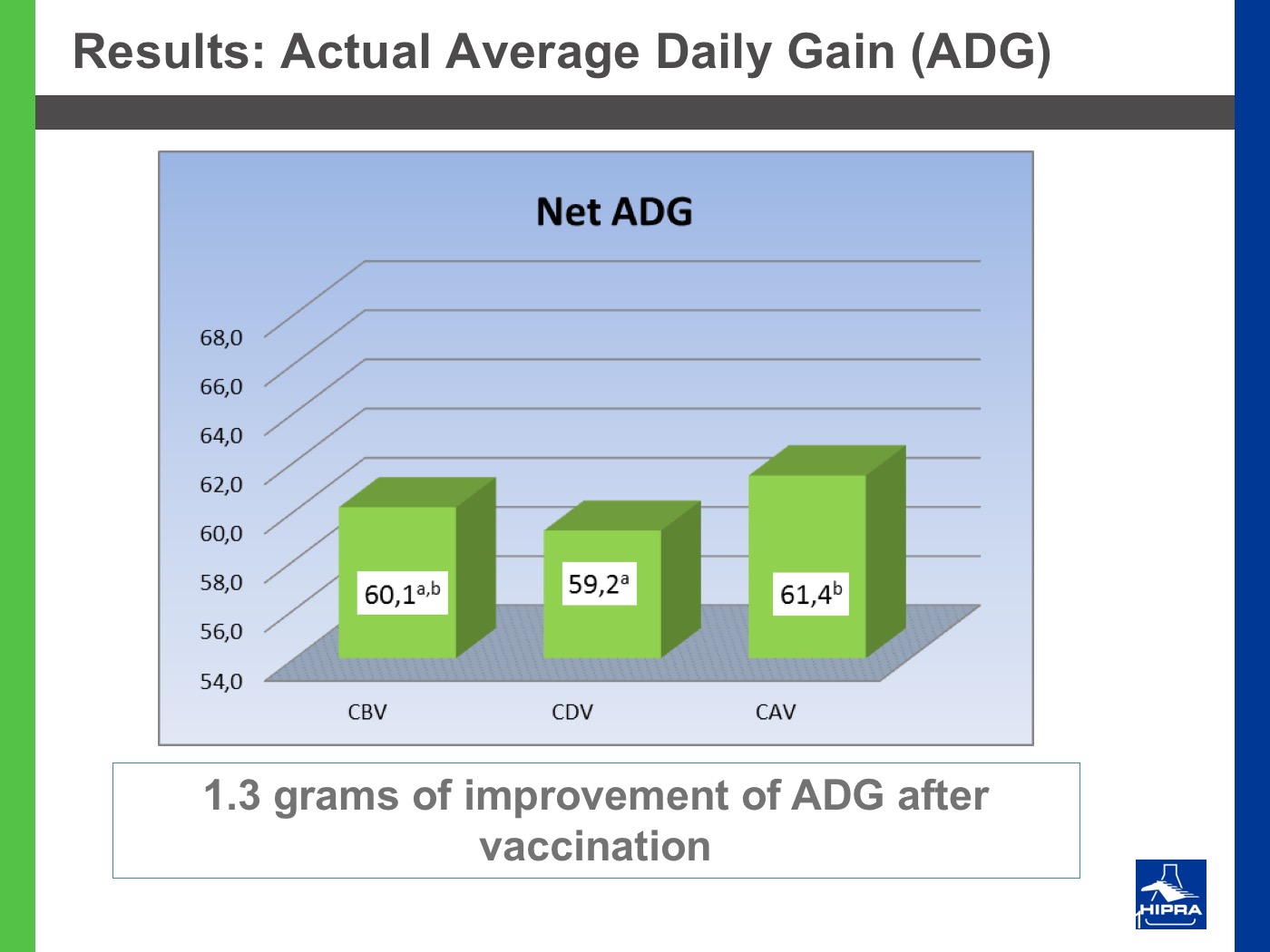
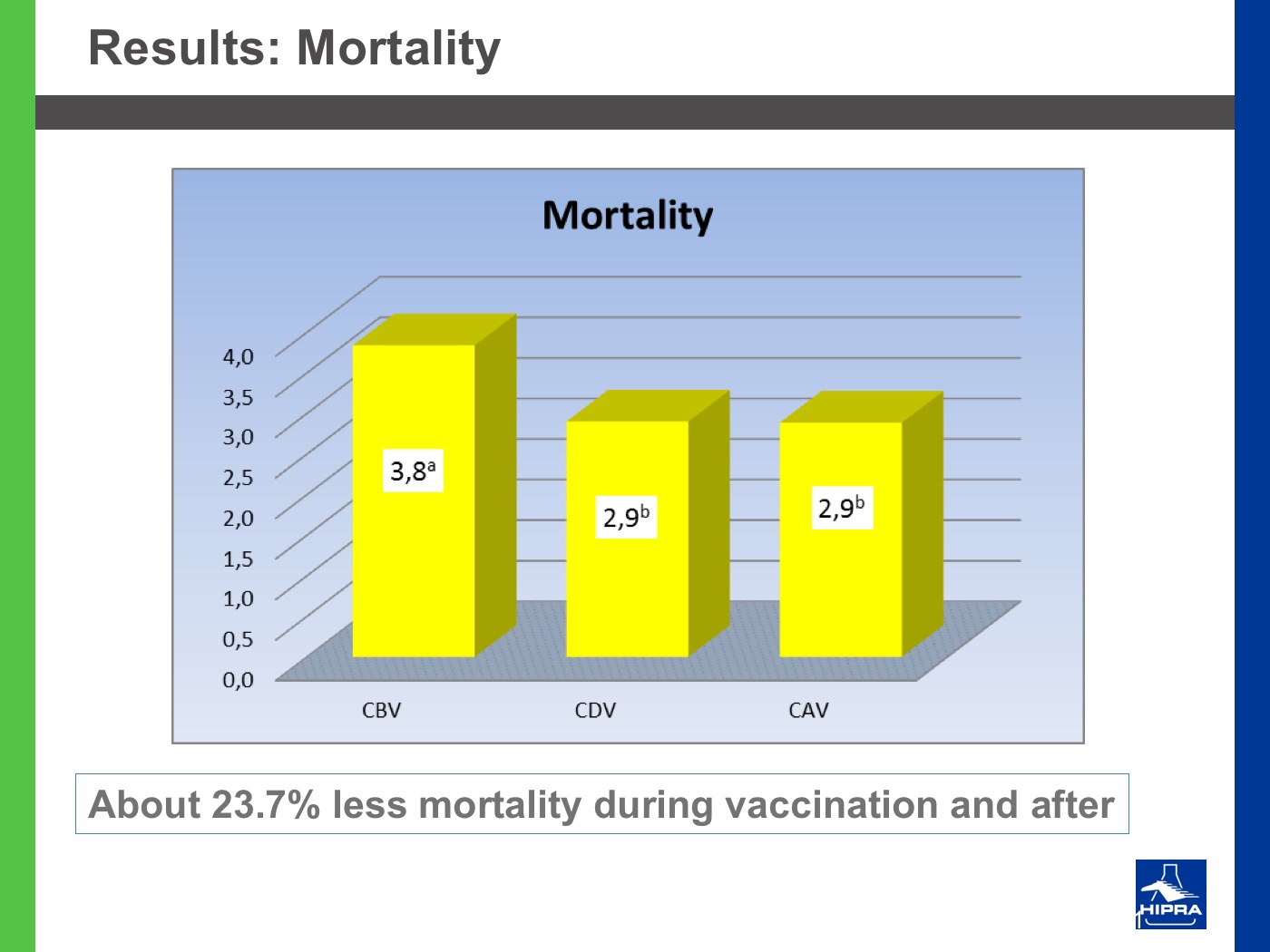
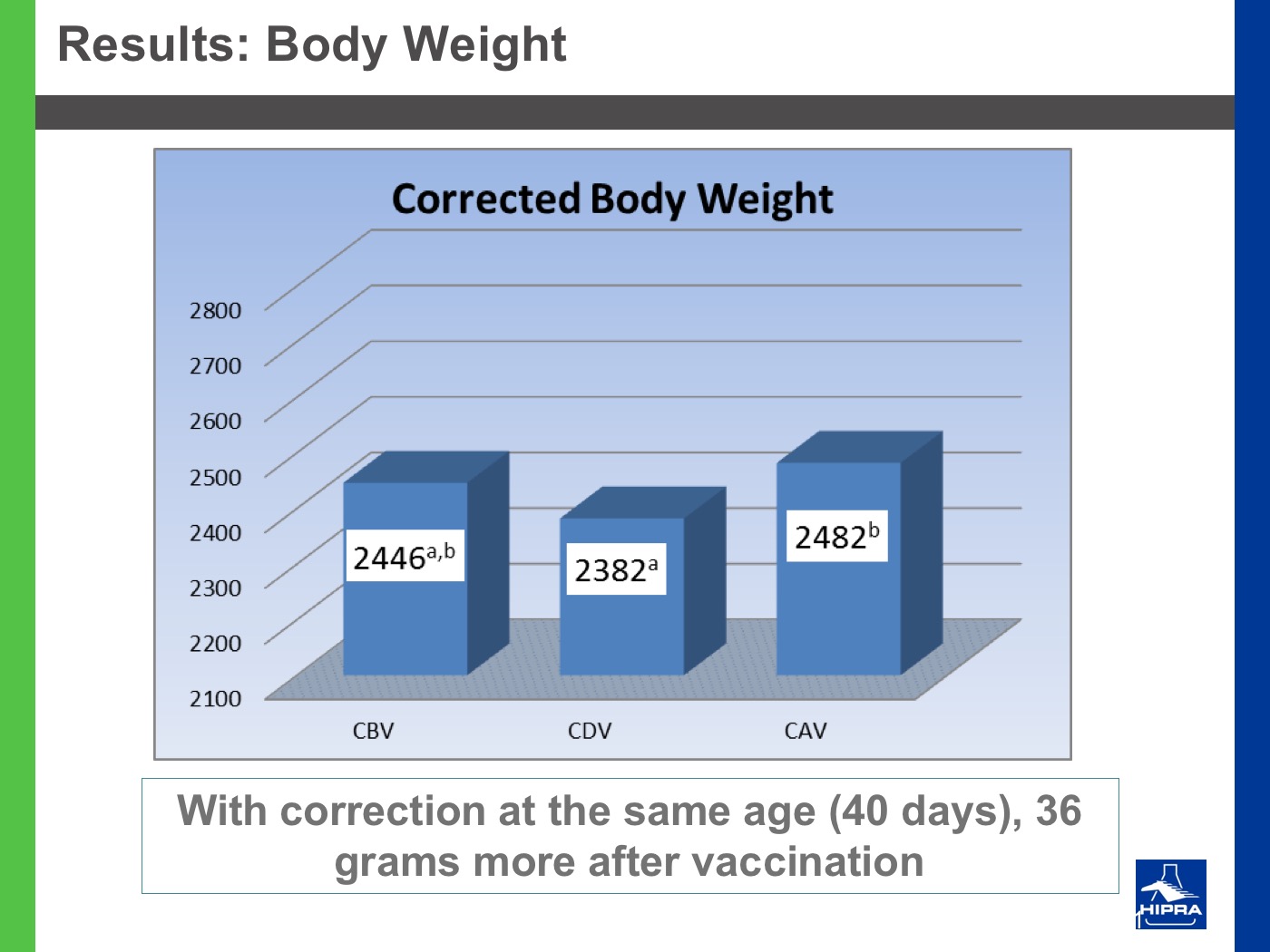
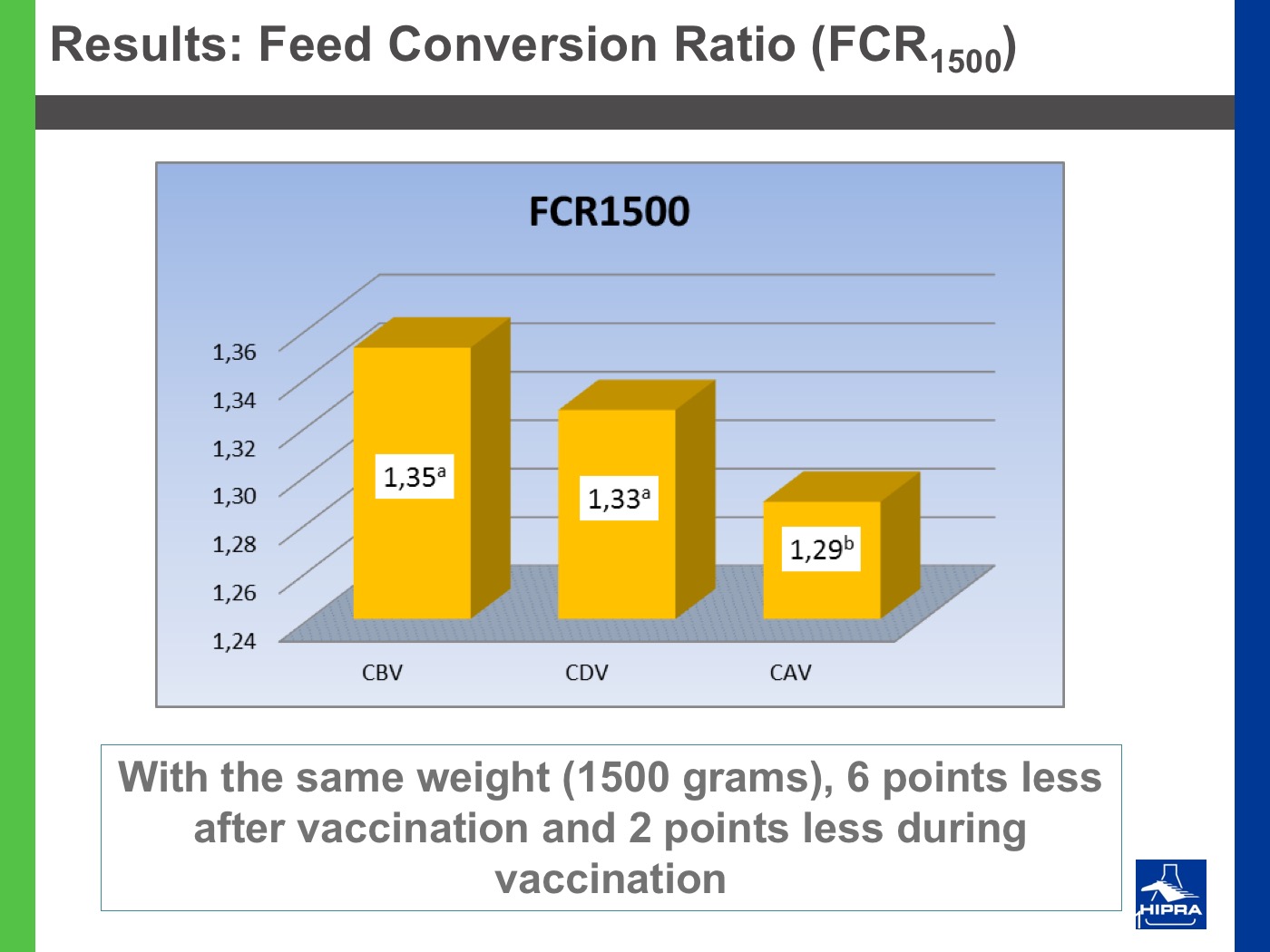
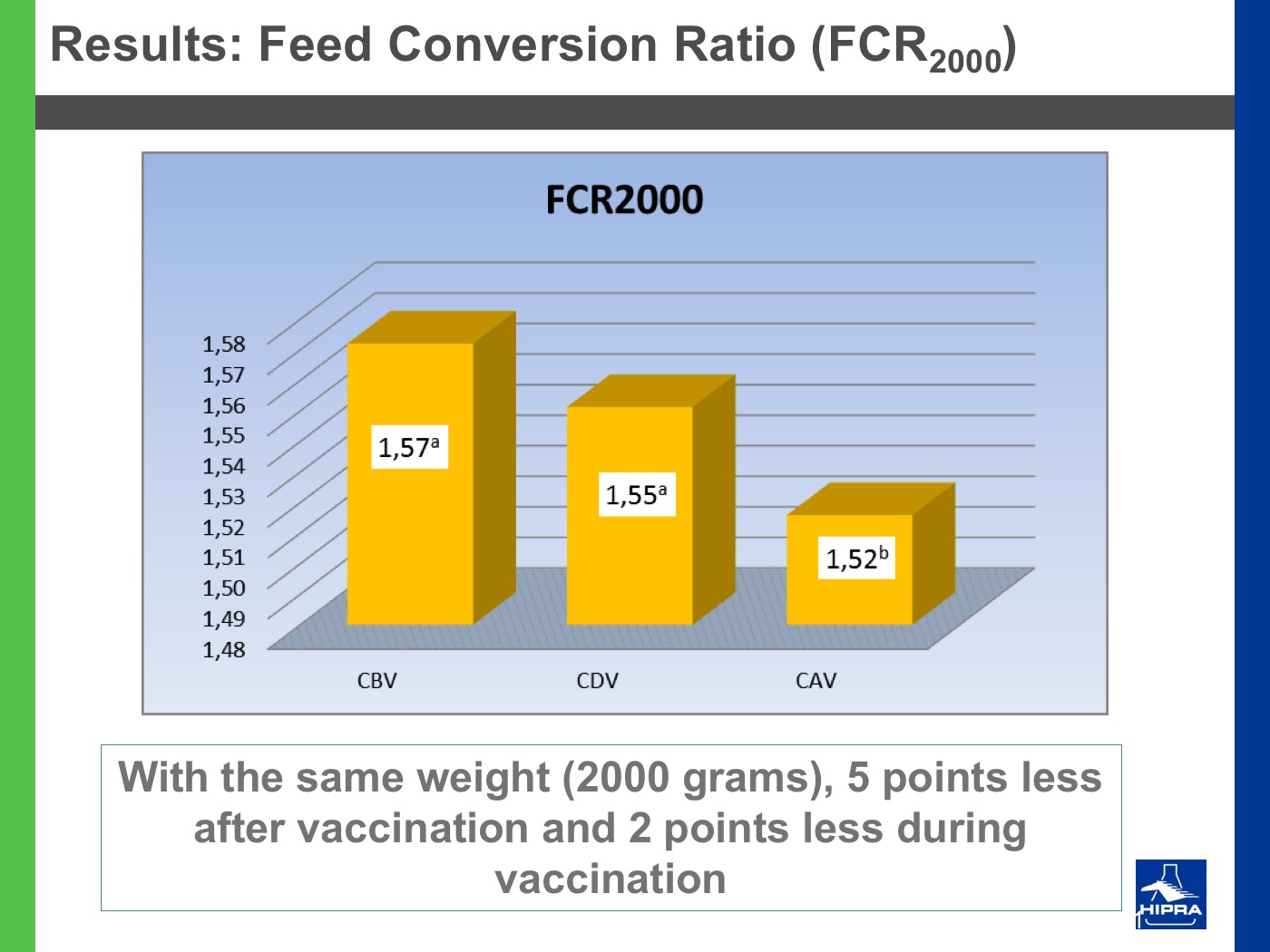
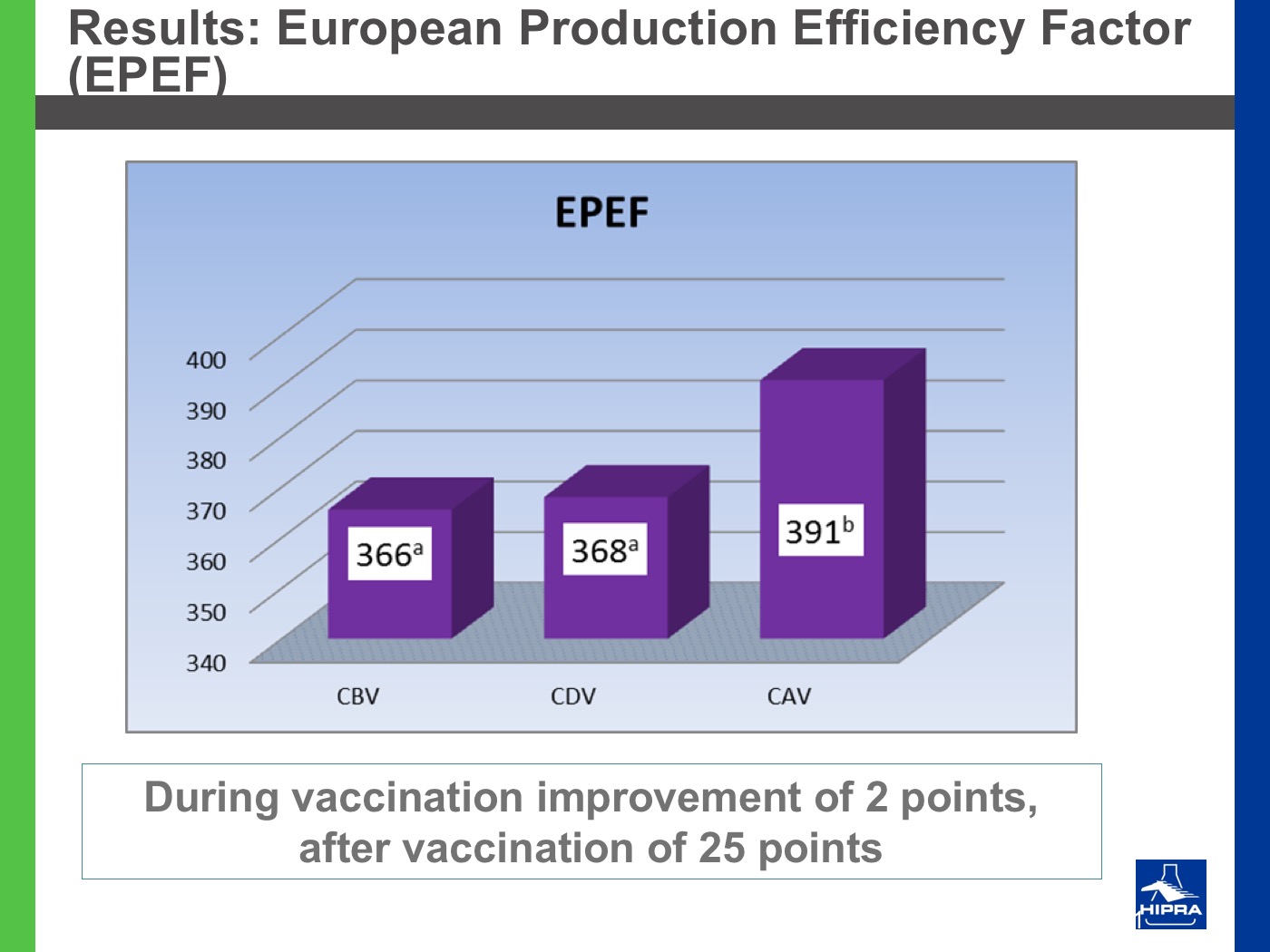
Nowadays, rotation programmes with Eimeria vaccines realistically represent the best long-term strategy to prevent coccidiosis in broiler productions but also to continuously improve production results, which is the ultimate goal for every poultry producer.
References
- Chapman H.D., 1997. Biochemical, genetic and applied aspects of drug resistance in Eimeria parasites of the fowl. Avian Pathology, 26 (2), 221-244.
- Chapman H.D., and Jeffers T.K., 2015. Restoration of sensitivity to salinomycin in Eimeria following 5 flocks of broiler chickens reared in floor-pens using drug programs and vaccination to control coccidiosis. Poultry Science 94, 943–946.
- Dardi M., Pagès M., Rubio J., Mathis G. F., De Gussem M., 2015. Anticoccidial Sensitivity test (AST) results from a farm vaccinated for three consecutive flocks with a coccidiosis vaccine. Proceedings of the XIX World Veterinary Poultry Association Congress. Cape Town, South Africa, 158.
- Marien M., De Gussem M., Vancraeynest D., Fort G., Naciri M., 2007. Indication of cross-resistance between different monovalent ionophores as determined by an anticoccidial sensitivity test (AST). 16th European Symposium on Poultry Nutrition, Strasbourg, France.
- Peek H.W., Landman W.J., 2011. Coccidiosis in poultry: anticoccidial products, vaccines and other prevention strategies. Veterinary Quarterly 31, 143-161.
- Ronsmans S., Van Erum J., Dardi M., Rubio J., 2015. The use of a live coccidiosis vaccine in rotation with anticoccidial feed additives: results from the Belgian field. Proceedings of the XIX World Veterinary Poultry Association Congress. Cape Town, South Africa, 158.
- Shirley, M.W., Harvey, D., 2000. A genetic linkage map of the apicomplexan protozoan parasite Eimeria tenella. Genome Res. 10, 1587–1593.
- Shirley, M.W., Smith, A.L., Blake, D.P., 2007. Challenges in the successful control of the avian coccidia. Vaccine 25, 5540–5547.
- Williams R.B., 1999. A compartmentalised model for the estimation of the cost of coccidiosis to the world’s chicken production industry. International Journal for Parasitology 29 , 1209 – 1229.