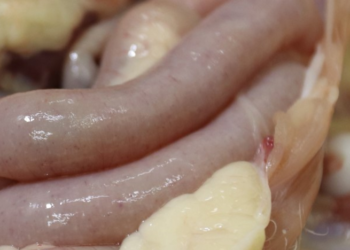
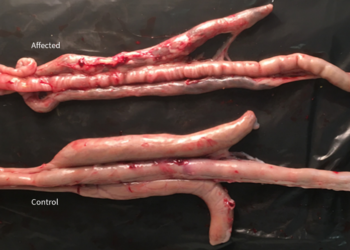
When talking about the diagnosis of Eimeria in chickens, most people think of the oocyst count (OPG) and now PCR as the gold standard methods for obtaining the most accurate diagnosis. This is partly true, however it is very important to remember that the earliest diagnosis is still only possible with the classical lesion scoring technique implemented in 1970 by Johnson & Reid. Some factors can affect the lesion scoring method and should therefore be taken into consideration: proper selection of birds, careful necropsy procedure and accurate training for lesion identification.
The oocyst count, commonly referred to as OPG (oocysts per gram of faeces), has been used for decades as the most common way to detect Eimeria oocysts on farms and it is still widely performed with the same purpose. However, we should point out its limitations:
- The presence of oocysts does not necessarily mean that a coccidiosis outbreak will soon occur on that farm;
- Affected birds with macroscopic lesions in the gut do not yet produce and shed oocysts.
In spite of this, the oocyst count has been shown to be very useful when monitoring the farm with weekly sampling, as the results can give us an indication of either the development of anticoccidial resistance or of the vaccine intake and onset of immunity after coccidiosis vaccination.
The PCR technique for Eimeria – recently introduced as a routine test in more and more diagnostic labs – also has advantages and limitations. The main advantage is the possibility of an accurate species identification of the Eimeria spp. contained in the sample.
In fact, field samples may contain debris and impurities and even if they are well preserved (refrigeration at 4-8ºC) and the typical morphology of each Eimeria species is easily recognizable, Eimeria spp. identification may be difficult.
Moreover, shape can be altered if the oocyst sporulates, thus an error range always has to be taken into account when species identification is done through microscopic appearance.
Whereas the main disadvantage of this technique lies in the fact that we do not know whether the Eimeria DNA that has been amplified comes from a live and infective parasite or from DNA debris contained in the sample.
This is why, the above having been said, lesion scoring for Eimeria in chickens today still remains the earliest method of diagnosis for coccidiosis outbreaks together with clinical signs. As there are some variables that can also influence this technique, in order to achieve the most reliable results, we should briefly analyse them:
1. Selection of the birds
Before necropsy, it is essential to know the Eimeria prevention strategy used on the farm: vaccinations or anticoccidials.
It has been shown that the existence of gut lesions is not necessarily accompanied by clinical signs of coccidiosis in poultry (Williams et al., 2000). It has also been shown that immunized birds may display lesions and high oocyst counts, with no growth retardation or clinically overt disease.
As for the number of birds to be examined, generally, a total of 5 to10 birds from each house would be reasonable. Regarding the age, unless in the presence of a coccidiosis outbreak, in the case of vaccinated birds, the best age to check the safety of the vaccine is between 18 and 24 days of age. On the other hand, efficacy of the vaccine is better checked by performing necropsy between 28 and 35 days of age.
If anticoccidials are used, the best age to check the efficacy of the compound used in the feed is between 25 and 35 days of age. These birds should be of the average weight of the batch, and be alive at the time of sampling, not debilitated or fasted.
2. Necropsy procedure for Eimeria lesions detection
As previously mentioned, it is crucial that birds examined for gross lesions (at least 5 per batch), are alive and within the appropriate age range. If multiple groups are inspected, it is better to sacrifice one group and then another to reduce post-mortem artifacts.
The intestine is the first organ to decompose after death. Postmortem changes negatively influence the assessment of pathological changes due to Eimeria infestations in the gut. Make the sacrifice humanely with the least possible stress.
3. Training for Eimeria lesion identification
To complete the inspection looking for Eimeria lesions in a systematic way, consider the segments of the intestine that are shown in the slide share below: duodenum, upper and lower mid-intestine, rectum and caecum.
Do not expose the inner surface of the intestine until you have inspected the outer wall. It is important to record any change in thickness, colour or presence of spots, either petechiae or white spots.
Expose the inside of the digestive system. Observe and describe the content found: amount and appearance. Inflammation of the intestine can sometimes be identified immediately after cutting the wall longitudinally, as it tends to fall back on itself.
This only happens when it belongs to a bird that has been recently sacrificed. List all the lesions observed, their location, colour, appearance, distribution, shape, etc. Whenever possible, take photos of the lesions.
Find some pictures and description of the typical lesions of the seven Eimeria spp. in the video above.
To sum up, in order to perform a careful coccidiosis diagnosis, we need to work with all three above-mentioned methods together and be well aware of the advantages and limitations of each.
References
- Williams R.B., Catchpole J., 2000. A new protocol for a challenge test to assess the efficacy of live anticoccidial vaccines for chickens. Vaccine 18(13): 1178-85.
- Johnson J., Reid W.M., 1970. Anticoccidial drugs: lesion scoring techniques in battery and floor-pen experiments with chickens. Exp. Parasitology 28(1): 30-6.