The Eimeria species responsible for coccidiosis in the species Gallus gallus are: E. acervulina, E. maxima, E. mitis, E. praecox and E. tenella, which are responsible for the disease in short life-cycle poultry (broilers), and E. necatrix and E. brunetti, which, together with the above 5 species, are responsible for the occurrence of outbreaks in long life-cycle poultry (breeders and layers). They are all ubiquitous in their behaviour and vary in their pathogenicity.
There are seven Eimeria species that are responsible for avian coccidiosis, 5 of which cause the disease in broilers: E. acervulina, E. mitis, E. tenella, E. maxima and E. praecox.
There are two more strains of Eimeria that are not recognised as causing the disease. These are: E. hagani, the only description of which was by Levine P.P. in 1938; and E. mivati, a species found in a vaccine in the USA which appears to be a mixture of other Eimeria, E. mitis and E. acervulina (M. W. Shirley et al. 1983).
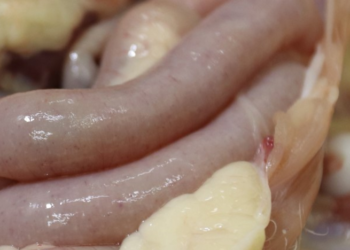
Focussing on the species that are really important in the generation of the disease in broilers, we need to know exactly which species cause most damage and how they interact by generating synergies between one another, thereby causing greater damage within the host. E. acervulina, E. maxima, E. tenella, E. mitis and E. praecox are the main species that cause avian coccidiosis in broilers. They can all be found along the intestine of poultry, affecting different areas and causing different lesions depending on the species concerned.
Focussing on E. acervulina, E. maxima, E. tenella, E. mitis and E. praecox, the species that are responsible for avian coccidiosis in broilers, we need to know the lesions they cause and their behaviour and distribution within the gastrointestinal tract of chickens. Below is a video giving details of the different species of Eimeria that affect the gastrointestinal tract of poultry.
For the development of vaccine strains, the Eimeria species have to be attenuated. There are three methods of doing this: passage through embryonated eggs, gamma irradiation and selection for precociousness. When they are selected by passage through embryonated eggs and by gamma irradiation, this is generally associated with a loss of immunity by the line and therefore stable attenuation is not maintained (Shirley M.W et al. 1984). The best method shown so far is selection for precociousness developed by Jeffers (1975). during the nineteen seventies. The method is based primarily on the reduction of the reproductive potential of the strain, resulting in attenuation of the virulence, maintenance of the immunogenicity and genetically controlled stability.
When the appropriate method of attenuating the Eimeria species has been identified, we need to know which species will be necessary for development of the vaccine. E. acervulina, E. maxima and E. tenella are well known as the pathogens responsible for the disease and are included in the great majority of existing commercial vaccines, but this is not the case with E. mitis and E. praecox, which have been regarded as species of “less importance” in the field.
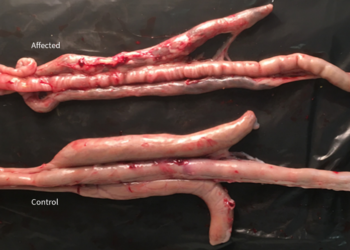
In his study on the pathogenesis of Eimeria praecox in broilers, R.B. Williams et al. (2009) demonstrated the importance of E. praecox as a pathogenic strain in itself. In this study, it was compared with E. acervulina and it was observed that the lesions caused by E. acervulina were macroscopic and severe, but did not last for more than 14 days post-infection. In contrast, in the case of E. praecox, the lesions were microscopic and caused a reduction in the viscosity of the intestinal content. In other studies such as those carried out by J.M. Répérant et al. 2011, the impact of the infection caused by E. praecox was related to the infective dose but its impact in performances was present from the lowest dose of 5000 oocysts/bird. When it was co-administered with E. acervulina, it caused a greater impact on production indices.
Furthermore, the selection of strains within an Eimeria species is essential in order to obtain good protection, thus conferring cross-protection within each species, as is the case with the vaccine strain E. maxima 013, which is able to provide protection against 6 different pathogenic strains of E. maxima obtained from different geographic locations.
It is essential to find out about the epidemiological behaviour of the Eimeria oocysts in order to understand how the vaccine strains will behave and what vaccine load will be necessary in order to produce an appropriate vaccine response. Generally, the oocysts that are found in the bedding can persist for up to 3 weeks (Williams R.B. 1995), with sporulation being better with a moisture content of the litter from 31 to 62.1%. Whether or not they are sporulated, 20% are ingested by the chickens and pass through their intestines. As immunity is generated within the flock, the percentage of oocysts eliminated is reduced with each life cycle of the parasite. Hence the necessity of determining accurately the volume of oocysts necessary for each vaccine strain, in order to ensure an appropriate response to the vaccine.
In order to confer immunity against all the Eimeria species that are present in the environment, all the Eimeria vaccine strains need to be included as there is no cross-protection between the different species, hence the necessity of formulating a vaccine with the 5 Eimeria species when the target category is represented by broilers.
With all this tested knowledge, HIPRA, a specialist in avian coccidiosis, has developed the HIPRACOX® vaccine, the only vaccine specially developed for short life-cycle poultry, containing in its formulation all the Eimeria species responsible for avian coccidiosis in broiler chickens.
REFERENCES:
- Jean-Michel Répérant, M. Dardi, M. Pagès, Martine Thomas-Hénaff., 2011. Pathogenicity of Eimeria praecox alone or associated with Eimeria acervulina in experimentally infected broiler chickens. Veterinary Parasitology 187, 333-336.
- B. Williams, R. N. Marshall, M. Pagès, M. Dardi and E. del Cacho, 2009. Pathogenesis of Eimeria praecox in chickens: virulence of field strains compared with laboratory strains of E. praecox and E. acervulina. Avian Pathology 38(5), 359-366.
- W. Shirley, T. K. Jeffers, P. L. Long, 1983. Studies to determine the taxonomic status of Eimeria mitis, Tyzzer 1929 and E. mivati, Edgar and Seibold 1964. Parasitology 87(2), 185-198.
- W. Shirley, M.A. Bellatti, 1984. Eimeria necatrix: Selection and characteristics of a precocious (and attenuated) line, Avian Pathology, 13:4, 657-668.
- Jeffers T.K. 1975. Attenuation of Eimeria tenella through selection for precociousness. J. Parasitology 61: 1083-1090.
- Levine P.P. 1938. Eimeria haganisp. (Protozoa: Eimeriidae) a new coccidium of the chicken. Cornell Veterinarian, 28: 263—266.
- Prowse S. J. 1991. Cell-mediated immunity to Eimeria in the fowl: The absence of cross-species protection is not due to the lack of cross-reactive T cells. International Journal for Parasitology 21(1): 133-135.
- Williams, R. B. 1995. Epidemiological studies of coccidiosis in the domesticated fowl (Gallus gallus). II. Physical condition and survival of Eimeria acervulina oocysts in poultry-house litter. Applied Parasitology 36(2): 90 -96.