Avian coccidiosis is a costly intestinal disease caused by parasites of the Phylum Apicomplexa, genus Eimeria, which frequently affect poultry. Apart from the three best known Eimeria spp. (Eimeria acervulina, Eimeria maxima and Eimeria tenella), other species can also affect chickens, and their appearance is mostly related to the category of birds under consideration: long or short-lived birds. In this post we will review the most important posts published so far on this subject and we will try to shatter a myth: do Eimeria mivati and Eimeria hagani really exist?
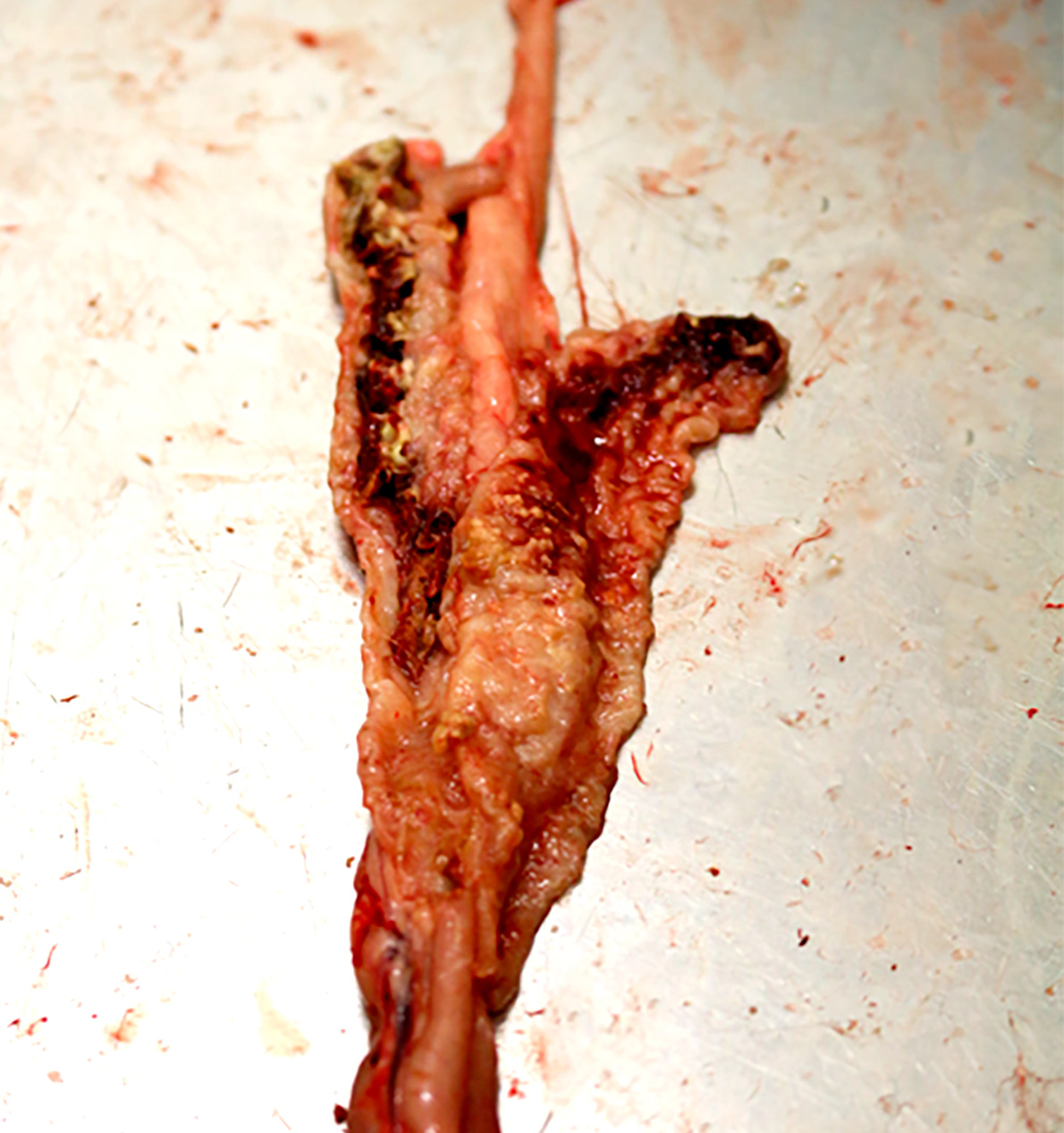 Eimeria tenella grade 4 lesion: caecal wall greatly distended with blood or large caseous cores; faecal debris lacking or included in cores (according to Johnson and Reid, 1970).
Eimeria tenella grade 4 lesion: caecal wall greatly distended with blood or large caseous cores; faecal debris lacking or included in cores (according to Johnson and Reid, 1970).
Chickens are susceptible to seven Eimeria species: Eimeria acervulina, Eimeria brunetti, Eimeria maxima, Eimeria mitis, Eimeria necatrix, Eimeria praecox and Eimeria tenella. Very often this is the start of many articles about coccidiosis, however many argue that another two species should be included in this list.
In previous posts we have already looked at which species of Eimeria are most involved, depending on the category of poultry we are considering. Focusing on broiler chickens we saw that the most relevant species are five in number, namely Eimeria acervulina, Eimeria maxima, Eimeria tenella, Eimeria mitis and Eimeria praecox.
At this point, it is worth mentioning that both Eimeria mitis and Eimeria praecox are usually considered to be subclinical species as they are not able to generate pathognomonic gross lesions in the gut, but they are able to affect productive parameters such as weight gain.
We all know that productive parameters are fundamental in broilers, as these birds have to grow quickly and as much as possible in a short period of time.
The role of E. praecox has been widely reviewed recently and it has been discovered that apart from being the cause of decreases in weight gain that cannot be recovered with compensatory growth (Williams, 2009), it is also able to create a synergism with Eimeria acervulina and have an adverse effect on weight and weight gain when compared to Eimeria acervulina alone, as both replicate in the same part of the gut, the duodenum (Répérant, 2012).
Moving on to long-lived birds such as breeders and layers, again five Eimeria species can lead to clinical coccidiosis in which the affected birds show typical symptoms of the disease, such as bloody droppings and increased mortality: Eimeria acervulina, Eimeria brunetti, Eimeria maxima, Eimeria necatrix and Eimeria tenella.
We can distinguish between caecal and intestinal coccidiosis. Caecal coccidiosis is due to Eimeria tenella that is confined to the caecum and consists mainly of the presence of haemorrhages on the inside of the wall of the caecum, followed by the formation of caseous cores in later stages.
This acute infection occurs most commonly in young chicks and is by far the most widely diagnosed in the field due to its typical lesions, localization and the fact that it is commonly associated with bloody droppings which are the first signs to alert the veterinarian or the farmer.
On the other hand, when talking about intestinal coccidiosis, this is usually caused by Eimeria necatrix (and less so by Eimeria brunetti). Both develop within deeper layers of the upper- and lower-mid intestine –Eimeria necatrix– and of the lower-mid intestine, rectum and caecum –Eimeria brunetti– and besides Eimeria tenella are major pathogens of long-lived birds.
In fact, both mainly affect birds of more than 8-9 weeks of age. Finally, Eimeria acervulina and Eimeria maxima develop in epithelial cells within the mid-intestine –Eimeria maxima– and upper-mid intestine and duodenum –Eimeria acervulina– and generally cause an intestinal coccidiosis more associated with marked weight loss and uniformity.
Furthermore, to continue with this brief survey of avian Eimeria spp., we have to mention two more species of Eimeria that nowadays are definitively recognized as nomina dubia.
These are: Eimeria hagani, which was described only once by Levine P.P. in 1938, after which no-one else was able to isolate it; and Eimeria mivati, a species which was found by one author to be a mixture of Eimeria mitis and Eimeria acervulina (M. W. Shirley, 1983), whereas Vrba discovered in 2011 that within its genome Eimeria mitis can carry two types of small ribosomal subunit (18S) gene that correspond to the known 18S sequences of Eimeria mitis and Eimeria mivati, and this conflicts with the existence of Eimeria mivati as an independent species.
Finally, to conclude, it is worth mentioning that in recent years three new cryptic Eimeria genomes -the so called operational taxonomic units [OTU] X, Y, Z- have been reported by some authors such as Cantacessi in 2008 in Australia and Clark in 2016 when he found that these three OTUs are widely distributed across the southern, but not the northern hemisphere.
Looking at the above, it is clear that the number of Eimeria spp. able to cause coccidiosis in chickens is far from being considered a closed subject.
REFERENCES:
- Cantacessi C., Riddell S., Morris G.M., Doran T., Woods W.G., Otranto D., Gassera R.B., 2008. Genetic characterization of three unique operational taxonomic units of Eimeria from chickens in Australia based on nuclear spacer ribosomal DNA. Veterinary Parasitology 154 (3-4), 226-234.
- Clark E.L., Macdonald S.E., Thenmozhi V., Kundu K., Garg R., Kumar S., Ayoade S., Fornace K.M., Jatau I.D., Moftah A., Nolan M.J., Sudhakar N.R., Adebambo A.O., Lawal I.A., Álvarez Zapata R., Awuni J.A., Chapman H.D., Karimuribo E., Mugasa C.M., Namangala B., Rushton J., Suo X., Thangaraj K., Srinivasa Rao A.S., Tewari A.K., Banerjee P.S., Dhinakar Raj G., Raman M., Tomley F.M., Blake D.P., 2016. Cryptic Eimeria genotypes are common across the southern but not northern hemisphere. Int. J. Parasitol. 46(9):537-544.
- Johnson J., Reid W.M., 1970. Anticoccidial drugs: lesion scoring techniques in battery and floor-pen experiments with chickens. Exp. Parasitol. 28(1): 30-6.
- Levine P.P., 1938. Eimeria hagani n. sp. (Protozoa: Eimeriidae) a new coccidium of the chicken. Cornell Veterinarian, 28: 263—266.
- Répérant J.M., Dardi M., Pagès M., Thomas-Hénaff M., 2012. Pathogenicity of Eimeria praecox alone or associated with Eimeria acervulina in experimentally infected broiler chickens. Veterinary Parasitology 187, 333-336.
- Shirley M.W., Jeffers T.K., Long P.L., 1983. Studies to determine the taxonomic status of Eimeria mitis, Tyzzer 1929 and E. mivati, Edgar and Seibold 1964. Parasitology 87(2), 185-198.
- Vrba V., Poplstein M., Pakandl M., 2011. The discovery of the two types of small subunit ribosomal RNA gene in Eimeria mitis contests the existence of E. mivati as an independent species. Veterinary Parasitology 183, 47-53.
- Williams R.B., Marshall R.N., Pagès M., Dardi M., Del Cacho E., 2009. Pathogenesis of Eimeria praecox in chickens: virulence of field strains compared with laboratory strains of E. praecox and Eimeria acervulina. Avian Pathology 38(5), 359-366.



